 
It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you” 1 (p. Rutherford described finding these results: “It was quite the most incredible event that has ever happened to me in my life. He shared the 1933 Nobel Prize for Physics with British physicist P.A.M. Pierre Curie carried a vial of radium in his coat pocket to demonstrate its greenish glow, a habit that caused him to become ill from radiation poisoning well before he was run over by a horse-drawn wagon and killed instantly in 1906.\)). Erwin Schrödinger (born August 12, 1887, Vienna, Austriadied January 4, 1961, Vienna) Austrian theoretical physicist who contributed to the wave theory of matter and to other fundamentals of quantum mechanics. Starting with several tons of pitchblende, the Curies isolated two new radioactive elements after months of work: polonium, which was named for Marie’s native Poland, and radium, which was named for its intense radioactivity. He performed experiments on radioactivity and is widely regarded as the Father of Nuclear Physics or Father of the Nuclear Age. She found that one particular uranium ore, pitchblende, was substantially more radioactive than most, which suggested that it contained one or more highly radioactive impurities. Ernest Rutherford was the first man to split an atom, transmuting one element into another. Marie Curie coined the term radioactivity (from the Latin radius, meaning “ray”) to describe the emission of energy rays by matter. Ernest Rutherford was a British physicist who discovered the nucleus of the atom in 1911 by firing alpha and beta particles at a thin gold foil. Although he earned his Nobel Prize in 1908 for the discovery of radioactive emissions, his greatest contributions came in the following years with the discovery of the nucleus and the proton of an atom. Becquerel’s work was greatly extended by Marie Curie (1867–1934) and her husband, Pierre (1854–1906) all three shared the Nobel Prize in Physics in 1903. Father of Nuclear Physics: Ernest Rutherford aptly earned the moniker of Father of Nuclear Physics for his contributions to the atomic theory. The second line of investigation began in 1896, when the French physicist Henri Becquerel (1852–1908) discovered that certain minerals, such as uranium salts, emitted a new form of energy. 
With this information and Thomson’s mass-to-charge ratio, Millikan determined the mass of an electron: 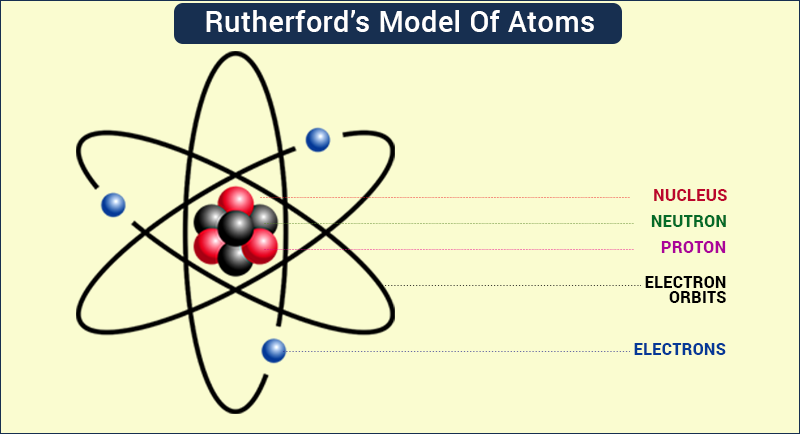
Subsequently, the American scientist Robert Millikan (1868–1953) carried out a series of experiments using electrically charged oil droplets, which allowed him to calculate the charge on a single electron. Another set of electrode plates deflect the ray, with the ray bending towards the positive plate. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Schematic of cathode ray tube with deflection. Thomson produced a visible beam in a cathode ray tube. (B) According to the plum pudding model (top), all of the alpha. Image used with Permission (CC BY-SA-NC). 2: (A) The experimental setup for Rutherfords gold foil experiment: A radioactive element that emitted alpha particles was directed toward a thin sheet of gold foil that was surrounded by a screen which would allow detection of the deflected particles. 
As the cathode rays travel toward the right, they are deflected toward the positive electrode (+), demonstrating that they are negatively charged. \): Deflection of Cathode Rays by an Electric Field. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
